The FDA and CDC, in collaboration with the California Department of Public Health (CDPH), Infant Botulism Treatment and Prevention Program (IBTPP), and other state and local partners, continue to investigate a multistate outbreak of infant botulism. Epidemiologic and laboratory analyses indicate that ByHeart Whole Nutrition infant formula is the source of this multistate outbreak of infant botulism.
Since the last update on December 10, 2025, reports of cases linked to this outbreak have slowed, and public health officials have not added any new cases to this investigation. Going forward, instead of weekly updates, CDC will provide updates on this outbreak investigation when new information becomes available. FDA will continue to provide investigational updates as needed.
All ByHeart infant formula products have been recalled, and these products should not be available for sale in stores or online. This includes all formula cans and single-serve “anywhere pack” sticks. On December 12, 2025, FDA sent warning letters to four major retailers for failing to remove recalled ByHeart infant formula from their store shelves despite being notified of the recall. On December 15, 2025, FDA issued a press releaseand reminded industry about its legal duties regarding food recalls under the Federal Food Drug and Cosmetic Act. FDA asked companies to follow best practices when carrying out recalls. This is especially important for recalls involving foods for infants and young children, who are among our most vulnerable populations.
Additional testing by ByHeart, FDA, CDC, and state partners is underway, and results are expected in the coming weeks. Positive sample results for finished product testing will be included and updated in the Sample Results section below.
FDA’s investigation is ongoing to determine the point of contamination. This advisory will be updated as information becomes available.
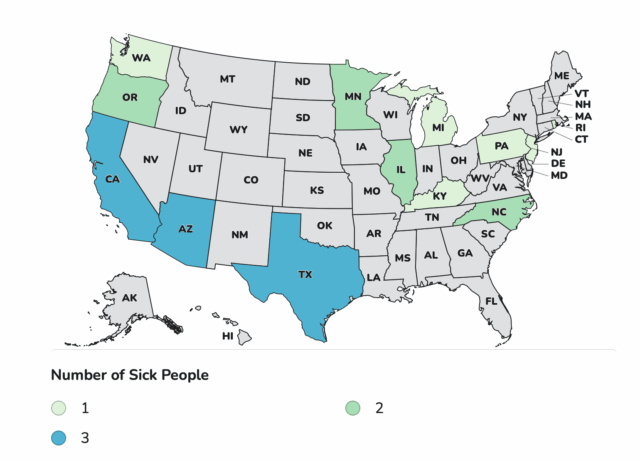
Case Count Map Provided by CDC

Case Counts
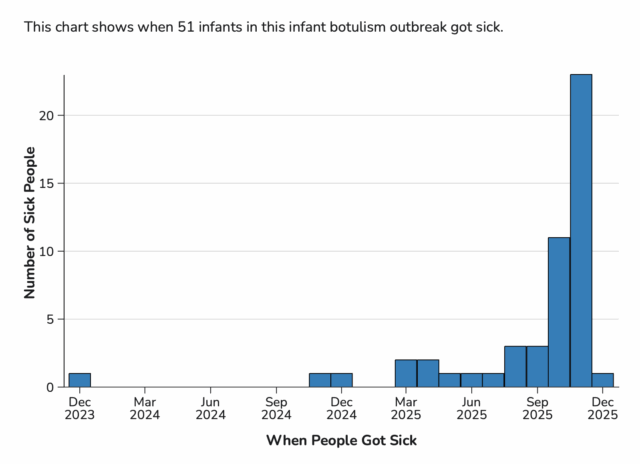
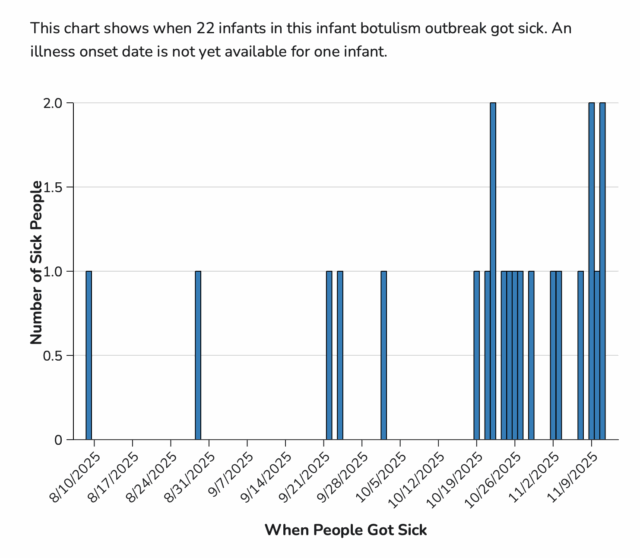
Total Illnesses: 51
Hospitalizations: 51
Deaths: 0
Last Illness Onset: December 1, 2025
States with Cases: AZ, CA, ID, IL, KY, MA, ME, MI, MN, NC, NJ, OH, OR, PA, RI, TX, VA, WA, WI
Product Distribution: Online and nationwide (including Guam and Puerto Rico), and internationally
Useful Links
- CDC Outbreak Advisory
- Infant Botulism Treatment and Prevention ProgramExternal Link Disclaimer
- ByHeart Expanded Recall Announcement
- Food Safety Tips for Retailers and Consumers During an Outbreak
- Food Safety Resources for Produce Shippers and Carriers During a Foodborne Illness Outbreak
- Who to Contact
- What is Botulism?